For the final class experiment I choose the experiment called Temperature and Heating Lab : Radiation and Color. The group leader explained that this experiment was done in order to see what color paint would be the most efficient in absorbing radiation and creating a low temperature for the household. This system would be used in the summer. To test out this theory the group brought four thermometers, markers in four colors, a lamp and a timer. What we had to do was color the outer shell of the thermometer case with the marker. Then we put them directly underneath the lamp for 20 minutes. The thermometers were set at 27 degrees. Afterward we checked the temperature to see which one had the lowest temperature and would be the most efficient. Going into the experiment, I hypothesized that the black thermometer would be the most efficient because it does not have a reflection. To my surprise the most efficient turned out to be the green thermometer. The black had a temperature of 42 degrees Fahrenheit and green had a temperature of 40 degrees Fahrenheit. turns out that green is in the middle of the color spectrum, therefore the properties of absorption and reflection are leveled to create a steady temperature. I never thought that a specific color could effect the temperature of a house. It was very interesting to me. I think that this concept could work, but it would have to be put into place, in an area of constant heat. Anywhere, that has a change in season would need a change in temperature. In winter the house would always be cold. Coal and other methods of heat production would be used. This would effect the environment and defeat the purpose of painting for a “greener” house. The good news is that it can be used in some parts of the world. Some areas have now started to paint their roofs with what are called “cooling colors”. These are green, red, and black. They have used different materials, like aluminum to cool down the houses. It is a very fascinating process, which is why I decided to try out this experiment.
Monthly Archives: December 2013
Fruit/Battery Experiment
 We utilize all sorts of batteries throughout our everyday lives. Cars, cellphones, and digital watches are all powered by some sort of battery. With that said, we were curious what kind of basic principles create a “battery.” A battery is a container that consists of one or more cells that produce an electro-chemical reaction when connected to a device. The experiment we chose was to create a battery out of a fruit (lemons, limes, oranges, bananas) using carbon as the constant element and switching out various conductive metals (zinc, copper, iron) as the second element. We hypothesized that the lemon would have the highest pH level and thus would generate the highest amount of voltage. Conversely, we had guessed that a banana would produce the least amount of voltage. For the experiment, we utilized a multi-meter to measure the voltage of each fruit and metal combination. Our results were fairly surprising. On average, copper was the least conductive metal – generating only .1 volts when implanted in a lemon. The galvanized (zinc-coated) nail produced the highest readings – producing .84 volts when implanted in an orange. The most surprising aspect of our findings was that the banana produced fairly high readings on the multi-meter. This is because the ascorbic acid found in a ripe banana tends to have a fairly higher pH level than the citric acids found in the other fruits.
We utilize all sorts of batteries throughout our everyday lives. Cars, cellphones, and digital watches are all powered by some sort of battery. With that said, we were curious what kind of basic principles create a “battery.” A battery is a container that consists of one or more cells that produce an electro-chemical reaction when connected to a device. The experiment we chose was to create a battery out of a fruit (lemons, limes, oranges, bananas) using carbon as the constant element and switching out various conductive metals (zinc, copper, iron) as the second element. We hypothesized that the lemon would have the highest pH level and thus would generate the highest amount of voltage. Conversely, we had guessed that a banana would produce the least amount of voltage. For the experiment, we utilized a multi-meter to measure the voltage of each fruit and metal combination. Our results were fairly surprising. On average, copper was the least conductive metal – generating only .1 volts when implanted in a lemon. The galvanized (zinc-coated) nail produced the highest readings – producing .84 volts when implanted in an orange. The most surprising aspect of our findings was that the banana produced fairly high readings on the multi-meter. This is because the ascorbic acid found in a ripe banana tends to have a fairly higher pH level than the citric acids found in the other fruits.

Brian Brewster, Kim Wallace, Joe Cesaro, Joe Scolley
Pandora’s Promise
Pandora’s Promise raises awareness about nuclear energy, its systems, and how it works in the past, present, and future. The movie outlines the importance of Nuclear energy which has being dramatize as something like a weapon against the world, and wants now the people to see that is less dangerous and more convenience in the fact of leaving fossil fuels which we see now how their are affecting strongly our environment.
The movie explains that in the next few decades, humankind will need to increase energy production as billions of people in the developing world lift themselves out of poverty and begin to live modern lives. Stressing the use of new energy to be clean and non-CO2 emitting to lower the risk of triggering a devastating global climate catastrophe is a main goal.
Robert Stone, a reporter for CNN, commented on the movie and its motives stating his opinions. Stone opinionated that Edwin Lyman, a scientist whom critically disagrees with the film, and others seem to suggest that nuclear energy is hopeless and that all research and development into its advancement should be abandoned, period, and that we should throw all of our efforts into renewables and energy efficiency alone. Given the overwhelming challenges we face in attempting to power a planet of almost 9 billion people with clean, affordable, non-CO2-emitting energy, that’s about the most irresponsible course of action imaginable.
Fukushima
First off, I’d like to address the fact that before being assigned this topic I was unaware of the Fukushima disaster and after speaking with roommates, friends, and colleagues, most are still unaware of the tragedy. Following a major earthquake in March 2011, a 15-metre tsunami disabled the power supply and cooling of three Fukushima Daiichi reactors. This natural disaster caused a nuclear accident on the 11th of that month. All three cores largely melted in the first three days. Radioactive releases continued to flow over days 4 to 6 eventually taking the accident rating from a 7 on the INES scale to a total of 940 PBq. After two weeks the three reactor units 1-3 were stable with water addition but no proper heat sink to remove decay heat from fuel. In the next few months, by July, they were being cooled with recycled water from the new treatment plant. reactor temperatures has fallen to below 80 degrees celsius towards the end of October. Apart from cooling, which wasn’t officially shutdown and announced till mid-december, the basic task ongoing was to prevent release of radioactive materials, particularly in contaminated water leaked from three units.
Since the accident of “3.11”, Japan has been trying to develop a new national energy policy; but even three years later has failed to adopt a new policy. While this may concern most it doesn’t come as a surprise given that developments in energy capacity and infrastructure are normally measured in decades, not years. Even as history shows, Japan is not the quickest to move on substance issues like this as it failed to do so in 1986 after Chernobyl. The implications of the Fukushima disaster continue to be felt around the world, particularly for the nuclear energy industry. Forty-eight of Japan’s 50 nuclear reactors remain off-line today. The NRA began revising safety standards over the summer of 2013, that the operators of reactors had complied with.
In conclusion I believe Japan’s energy challenge over the next several years should indicate setting a clear energy for the future, and building strategies to execute the vision exploiting Japan’s world-beating technologies to benefit the people of its own islands, future generations of Japanese.
References:
http://worldnews.nbcnews.com/_news/2013/10/03/20797895-water-6700-times-more-radioactive-than-legal-limit-spills-from-fukushima?lite
http://en.wikipedia.org/wiki/Fukushima_Daiichi_nuclear_disaster
http://www.theguardian.com/environment/2013/dec/03/fukushima-daiichi-tsunami-nuclear-cleanup-japan
Solar Energy
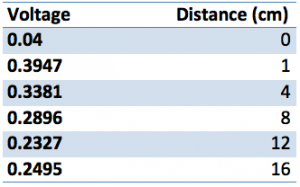
In class we partook in a small solar experiment in which we connected a small solar cell to the NXT adapter to measure the cells voltage with a probe. Placing a flashlight at varying distances from the solar cell we collected data aligned with each attempt. The chart below exhibits that the voltages acquired by the solar cell gradually decreases as the flashlight is distanced further away.
Placing for different colored film filters over the flashlight for the specific distances measures how different light spectrums affected the voltage of the solar cells. 
After conducting the experiment in the same manner as we did for our first data collection, we came across these results for the colored filters;