For our final project, we created a coffee cup experiment that measured which common coffee cup insulates heat the best and for the longest amount of time. We were curious about which material would insulate the best because, since millions of people in the world drink coffee or some other hot beverage daily, we wanted to see which material actually worked the best and was the most efficient. The three most common materials used to carry around a hot beverage seem to be a plastic travel mug, a paper mug, and a styrofoam cup. We decided that these were going to be the three containers that we would test.
Our theory, or hypothesis, before we ran the experiment was that the plastic travel mug would insulate the best because that is what they are designed to do. These mugs are reusable, popular, and seem to hold heat for a fair amount of time. We knew from the beginning that the disposable paper mug was not going to preform well, so we were mainly interested in the results from the plastic travel mug and the styrofoam cup. Also, based on our own experiences purchasing hot drinks over time in each of these different materials, we thought that either the styrofoam cup or the travel mug would preform the best.
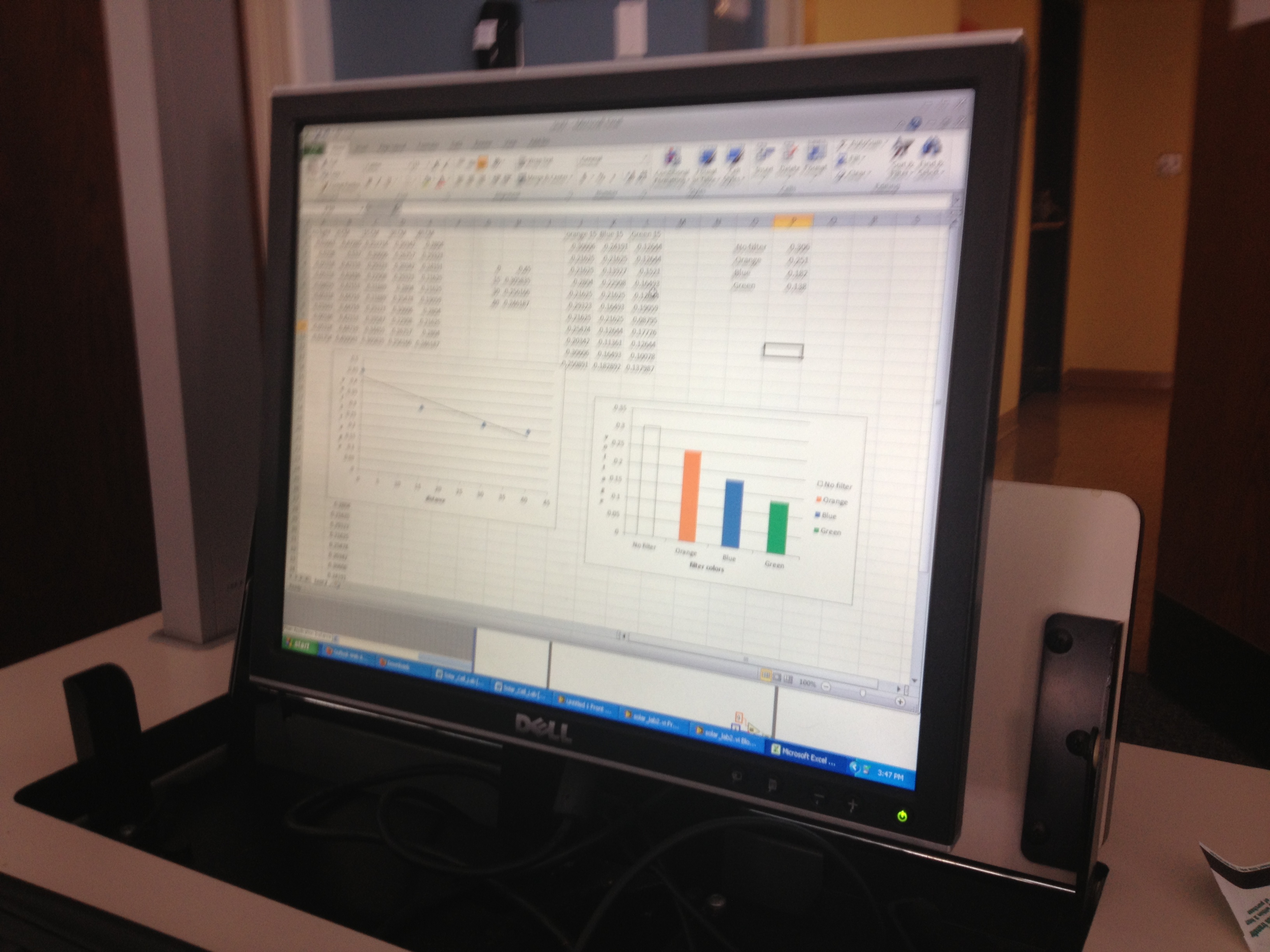
For this experiment, we didn’t need a lot of materials; We used: 1 NXT brick, 1 paper mug (with lid), 1 styrofoam cup (with lid), 1 plastic travel mug (with lid), 3 NXT temperature sensors, LabView computer software, hot water, and a permanent marker to mark where the water line was. First, we set up the LabView and NXT brick with the adaptors to the computer. Then, we marked a line on each cup to where we were going to pour the hot water in. Next, we poured hot water into each cup up to the line that we marked. Finally, the temperature probes go in, and the lids go on the containers. Over the next 15 minutes we measured the temperature decrease of each container every 10 seconds. This data was captured in the LabView program, and subsequently transferred to an Excel Document. At the end of the experiment, all of the temperature data is collected and the water is disposed of and everything is put away.
Our results for this experiment were fairly on par with our hypothesis in the beginning. The paper mug did in fact lose heat the fastest. The starting temperature for the hot water was 158 Degrees Farenheit, and the paper mug’s temperature rapidly fell. However, we were surprised to find out that the styrofoam cup insulated the heat better than the plastic travel mug. Since travel mugs are designed to insulate, we were shocked to see that the inexpensive styrofoam cup hold out the longest.
Overall, we are hoping that the students performing this experiment will gather similar results that we did, that will benefit owners of coffee shops. Since everyone is trying to make themselves and their businesses more efficient, these results will be important for them when it comes time for them to purchase cup materials. We also hope that the students can see the relationship between this experiment and renewable energy. Having an efficient product that can insulate heat energy best is important. In addition to that, styrofoam is not good for the environment, while paper mugs can be recycled and travel mugs reused. We hope that students can come to a conclusion on whether stores should invest in a cheap, efficient product that has a greater negative effect on the environment, or choose the less efficient product that can be reused/recycled. In the end, the group of students did successfully gather results that were similar to ours, and the understood the purpose of the experiment.
Below is a copy of our handout that the other group received.

COFFEE CUP EXPERIMENT
Purpose: To determine which container best insulates heat energy the longest.
Background: This particular experiment will help coffee shop owners decide which type of container will keep their product insulated for the longest amount of time for its customers. In the exhibition, hot water is placed into three types of coffee containers. The containers include a Styrofoam cup, plastic mug and paper cup. Thermometers collect the temperatures of the liquid in each cup over the course of a half hour. Keep in mind that the water started at an equal temperature and equal level in all three containers. Every ten seconds a temperature is recorded. Efficiency and sustainability is very important these days, and learning which container best insulates will help that cause. Using a container that best insulates heat will cut down on energy to re-heat the beverage, while also keeping your beverage warm.
Procedure:
Setup: Collect your materials: 1 paper mug (with lid), 1 styrofoam mug (with lid), 1 plastic travel mug (with lid), 1 NXT Brick with 3 adaptors cords, 3 NXT temperature probes, hot water, LabView software, and a permanent marker.
Mark a line on each container to where the hot water will be poured in. Next, set up the NXT brick with the cables and sensors and open up the LabView software. Then, pour in the hot water up to the marked line and put in the NXT temperature probes. Finally put the lids on (as best as you can) to each container and begin to gather your results.
Data Collection: The NXT temperature probes will be constantly measuring the temperature of the hot water inside each cup. This experiment will run for 30 minutes, with the sensors gathering temperature data every 10 seconds. After you have all of your data in the LabView software, open up an Excel Document, and begin to record/graph your data.
Data:
| Container At Starting Temperature (F) | After 10 Seconds | After 30
Seconds |
After 1 Minute | After 5 Minutes | After 10
Minutes |
After 20
Minutes |
After 30
Minutes |
| Travel Mug: | |||||||
| Styrofoam Cup: | |||||||
| Paper Mug: |
Analysis: 1. Which container insulated the heat the longest?
2. Which container lost heat the fastest?
3. Which container material would you suggest to a store owner? Does the material and it’s relationship to the environment affect your decision?